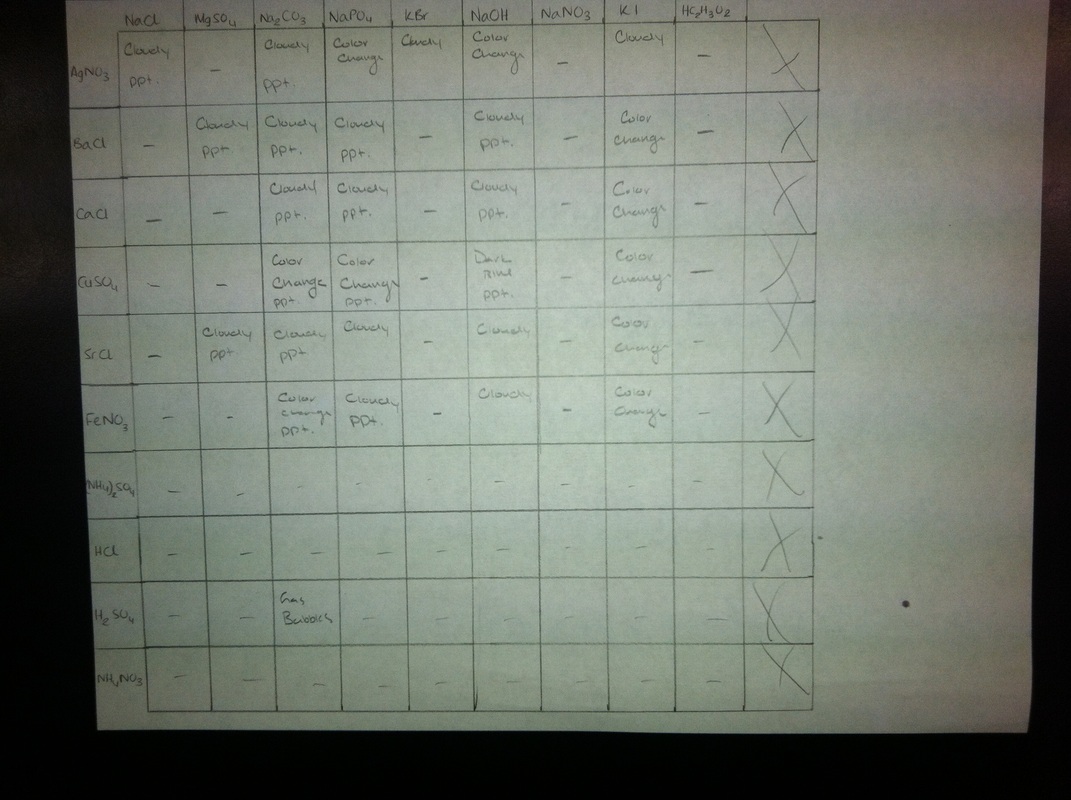
Data Table:
Conclusion:
Through the solubility rules, you can observe reactions between different solutions. Some reactions occurred because one of the products was not aqueous. Other mixtures did not show a reaction because both of the products of the reaction were aqueous.
Discussion of Theory:
The major theme of this lab was learning and understanding the solubility rules. Many double replacement reactions may not work because they form two aqueous products. This means that ions are just floating around instead of forming a chemical change. Other reactions do involve chemical changes. You can tell a reaction occurs because the solution may become cloudy, bubbles are formed, there is a color change, or a precipitate (solid) is formed. The reactions that we could measure in this lab all had one of these changes occur. The reason that these changes occur is because some of the solutions have stronger bonds than others. If the compound dissociates into ions then they will just float around. If the forces are not as strong then the products will stay together and not separate into ions. Instead they will form a precipitate. As seen in the picture above many of the solutions turned white or cloudy showing that a precipitate was forming. When sulfuric acid and sodium carbonate were mixed they showed a different chemical change. Gas bubbles were formed. When sodium phosphate and silver nitrate were mixed together a color change occurred. Both solutions were clear when in their original bottles but then when a drop of each were mixed it formed a yellow solution. We then used the data to determine the reactions that occurred. Using the reactions we could come up with the net ionic equation for each of the reactions. This means that we took out the extra ions that were not involved in the reaction and only included the important pieces that formed a solid or another change was noticed.
Sources of Error:
- Accidental mixing of more than two of the solutions
- Color change from one reaction spread into other solutions showing a false sense of a reaction occurring
- The solutions could have been contaminated to start with
Analysis Questions:
1. Some general statements about the solubilites of the ions are all sodium and potassium compounds are soluble, all nitrates are soluble, barium compounds usually are not soluble and silver compounds usually aren't soluble.
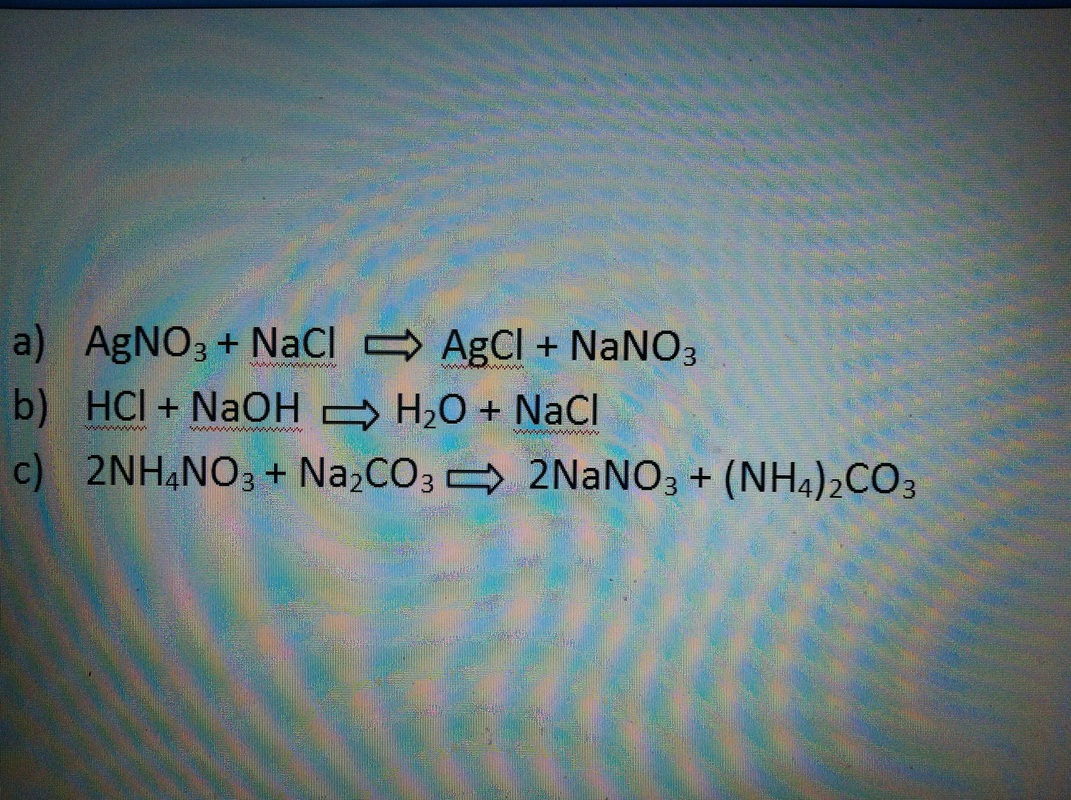
2. In picture below
2. In picture below
3. a) Formation of precipitates- two soluble ionic compounds are mixed together and an insoluble product is formed resulting in the precipitate
b) Neutralization reaction- acids and bases combine to form salt and water
c) Evolution of gas metastasis reaction- a non soluble gas is formed
-all examples in picture below
4. Water is a polar molecule. When split up into ions it splits into positive and negative ions because its unequal sharing of electrons. When something is dissolved in water ions with negative charges will bond to the hydrogen and ions with positive charges will bond to oxygen. If solutions are strong then they will split up into the ions but if they are weak then they will stay in their compounds.
5. Aqueous products are just individual ions floating around in water.