Procedure:
- Measure the mass of the lighter in grams and record
- Fill the bucket with water
- Measure the temperature of the water
- Using the sheet given, find the pressure of the water vapor for the temperature recorded
- Measure the mass of the empty jar
- Fill the jar with water as close to the top as possible and measure the mass
- Put the jar under water and take off the lid
- Put the lighter under water and press the button to allow the gas to go into the jar, making sure the bubbles only go in the jar
- After filling the the jar 2/3 with gas, put the lid on and remove the jar
- Weigh the jar in grams
- Allow lighter to dry and then weigh it
- Calculate the pressure of the gas by subtracting the pressure of the water vapor in atm from 1 atm
- Subtract the jar with gas from the only water jar to find the volume of the gas
- Subtract the new weight of the lighter from the initial weight
- Convert the temperature of the water from Celsius to Kelvin
- Use pv=nrt to find the moles of the gas
- Divide the number of grams of gas by the moles of gas to get molar mass
Data:
Mass of jar with only water: 462.7g
Mass of jar with gas & water: 369.8g
mL of gas: 92.8 mL
Temp of water: 22 degrees Celsius/295 degrees Kelvin
Pressure of water vapor: .026 atm
Pressure of gas: .974 atm
Initial mass of lighter: 16.9 g
Final mass of lighter: 16.7 g
Grams of gas used: .2 g
Moles of gas: .0037 moles
Molar mass of gas: 53.59 g/mol
Mass of jar with gas & water: 369.8g
mL of gas: 92.8 mL
Temp of water: 22 degrees Celsius/295 degrees Kelvin
Pressure of water vapor: .026 atm
Pressure of gas: .974 atm
Initial mass of lighter: 16.9 g
Final mass of lighter: 16.7 g
Grams of gas used: .2 g
Moles of gas: .0037 moles
Molar mass of gas: 53.59 g/mol
Calculations:
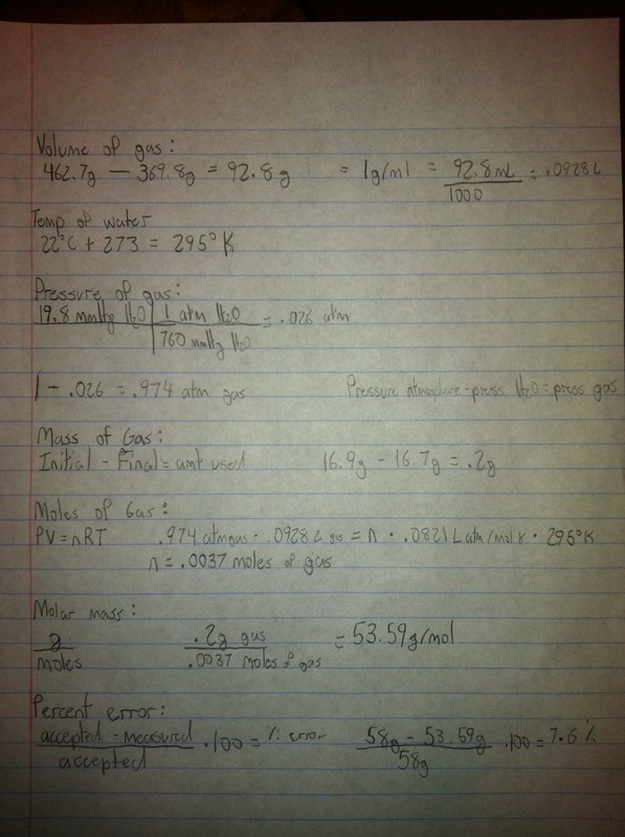
Conclusion:
By finding the amount of moles of gas by using the pv=nrt equation, we could divide that into the grams of gas used to find the molar mass. The gas was not 100% butane so that is why the molar mass varies by a little from the molar mass of butane.
Analysis:
- Based on our calculations, the molar mass of the gas is 53.59 g/mol.
- With our molar mass of 53.59 g/mol, a possible formula for the gas could be C3H17.
- Our percent error for the lab is 7.6%. Work is in picture above.
- a. This would give you a lower temperature. By dividing by a smaller number would give you a larger number of moles. If there is a larger number of moles, the molar mass will be lower because you are dividing by a larger number.
c. If a heavier gas is already present that will make it seem like the gas is heavier. This will result in a higher volume of gas and therefore a higher number of moles of gas. A higher number of moles yields a smaller molar mass.
d. This will result in a higher mass of lighter left after the experiment was performed. This means less you will have a smaller number of gas used. When dividing there will be a smaller number for grams which will result in a smaller molar mass.
5. The other gasses have a smaller mass. This can be concluded because if the gas was 100% butane the molar mass would be 58g/mol. Our number was smaller than 58 therefore the gasses must also be smaller in molar mass.
6. Butane would normally be a gas at standard pressure and temperature. Therefore the butane is a liquid because it is kept in very high pressure in the lighter and then when it is released into standard pressure a phase change occurs and it now becomes a gas.
